|
The majority of people, nonetheless, referred to it as a oganesson. Oganesson's finding in Russian federationīefore, this aspect has received numerous titles, including eka-radon and ununoctium. Instead, they kind diffuse blobs of cost that appear to be smeared out. The atomic composition of oganesson is very unusual, as the electrons are spread uniformly and you should not type distinct shells. Oganesson is really a new superheavy factor, discovered from a team of nuclear physicists and theoretical chemists from Michigan Condition University and Massey School in New Zealand. Its official name will not be adopted until after that period is over, though in the meantime, the element is known as tennessine. The new label will probably be completed by IUPAC following a five-calendar month overview time period. This new factor swithces the temporary systematic brand, ununseptium. Its atomic quantity is 117, symbolizing its chemical factor reputation. Tennessine is a recently discovered aspect in the routine kitchen table. These factors share similar chemical and actual components. /caesium-cs--chemical-element--3d-rendering-isolated-on-black-background-922704282-5adb6de08e1b6e0037170151.jpg)
A chart in the electron styles of all components demonstrates a fascinating pattern: aspects with one particular electron in the outermost orbital are situated in line one among Mendeleev's routine table. Mendeleev initial reported this regulation in 1869, along with the Pauli exclusion basic principle offered crucial theoretical assist with this idea. The periodic law states that compound aspects have a typical style of properties. The Most Reactive Elements On The Periodic Table. You can also learn about the IUPAC, which has recently reviewed elements 117, 113 and 115 and 118 and assigned them official symbols and names. These new elements need to be assigned a temporary symbol and name before an official symbol and name can be assigned. For instance, search for about the Pauli Exclusion Concept, Oganesson, or perhaps the discovery of the factor tennessine in Russia. Finally, fluorine has higher reactivity than chlorine.The Most Reactive Elements On The Periodic Table - There are several approaches to find a new Periodic Dinner table factor. Because fluorine’s atomic size is smaller than chlorine’s, the force of attraction between the nucleus and the valence electrons of the valence shell is stronger in fluorine, allowing it to obtain one electron more easily. As we proceed from top to bottom, the reactivity of the non-metallic elements in Group VIIA declines. Why is fluorine more reactive than chlorine: Fluorine and chlorine are nonmetals belonging to the periodic table’s Group VIIA. Because helium has an outer electron shell filled with electrons. 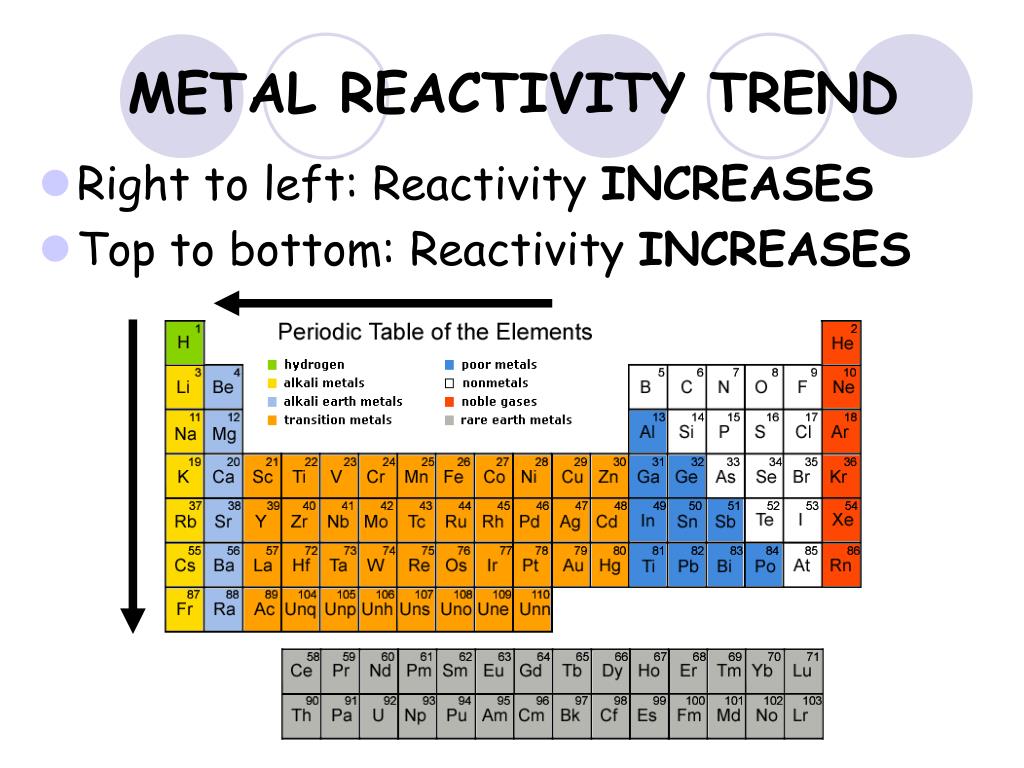
Helium is the 2nd most abundant element in the universe at room temperature. Helium (It is a Nonmetal) is the least reactive. Gold, silver, copper, carbon, nitrogen, oxygen, fluorine, and iron are examples of nonmetals.  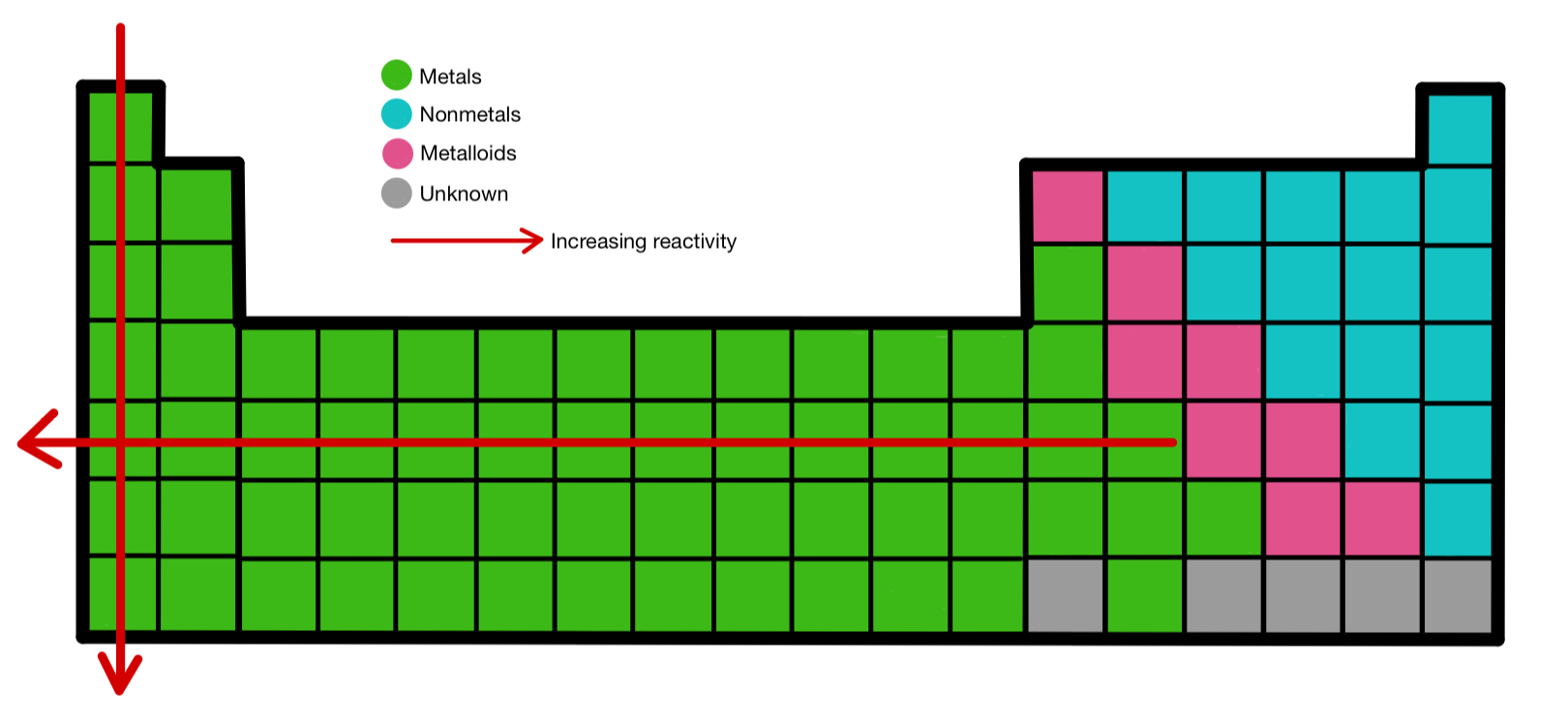
The periodic table can explain how elements are categorized and how they are related to one another.Ī chemical word for any element that does not have a metallic is a nonmetal. The periodic table is a diagram that displays chemical elements in order of atomic number. It reacts with water, forming hydrofluoric acid and hydrogen fluoride gas: HF + H 2O → H⁺ + HF + OH⁻ Fluorine has a boiling point of -188 ☌ (-310 ☏). Fluorine is a highly reactive, diatomic gas that can react with almost every other element and combine to create compounds with properties that are radically different from either starting ingredient. It has the highest electronegativity of any element, 3.98 on the Pauling scale. The most reactive nonmetal in the periodic table is fluorine. What is the most reactive nonmetal in the periodic table?
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
